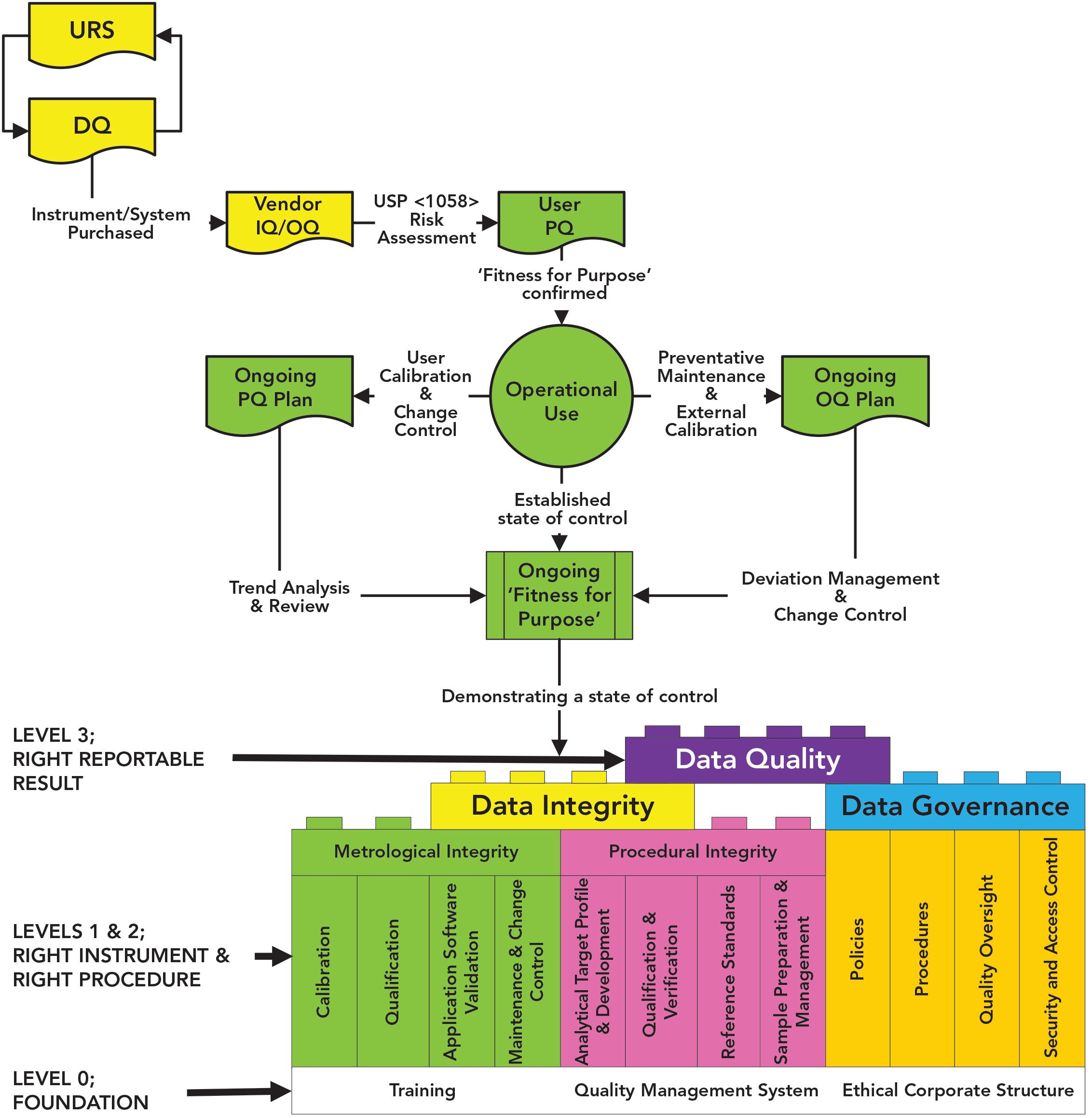
GAMP 4 AND GAMP 5 GUIDELINES. VERIFICATION
gamp 4 and gamp 5 differences Key role of the suppliers Ensuring compliance and fitness for purpose is the responsibility of the regulated company The supplier gets more liberties, but also more responsibilities The supplier gets the liberty to carry out the system development in accordance of accepted development- and quality management methods ( ISO 9000) without consideration of specific GxP requirements Documentation of the suppliers can be taken directly and without changing the format in the validation documentation of the regulated company (no duplication)ĥ Services of the suppliers should be overtaken considerable as possible (in specification and verification processes) Slide 111. But also the classical nomenclature can be used Unification of the following Guidelines:-FDA: Pharmaceutical cGMPs for the 21st Century: A Risk based Approach-ASTM E2500- gamp 5-ISPE Baseline-Guide Commissioning and Qualification Slide 101. Benefit of the risk based approach: Better knowledge about the processes and because of that a better process control (also described in ICH Q8 (QbD), ICH Q9 (QMS), ICH Q10 (PQS)) Safeguard patient safety, product quality, and data integrity, while also delivering business benefit A central element to reducing the validation effort by concentrating on the most critical processes and therewith reducing the validation costs Slide 91.Ĥ gamp 4 and gamp 5 differences Aligned terminology The classical nomenclature of IQ, OQ and PQ are not longer used in gamp 5 Implementation of the word verification instead of the classical nomenclature Focusing of the contents of the created documents not longer of the nomenclature. Section 5 describes this approach and how the activities should be based on good science and product and process understanding.

It is an iterative process applied throughout the entire system life cycle. gamp 4 and gamp 5 differences Quality Risk Management (QMS): The QMS is a systematic approach for the assessment, control, communication and review of risks to patient safety, product quality and data integrity. gamp 4 and gamp 5 differences Life Cycle Phases Slide 81. gamp 4 and gamp 5 differences Life Cycle The general simplified Life Cycle: Slide 71. gamp 4 and gamp 5 differences Overview of the significant changes gamp 4 to gamp 5 gamp 5 now describes the whole life cycle (including: concept, project, operation, retirement) gamp 5 arranges the Quality risk management as a central element in all phases of the life cycle gamp 5 was aligned with the ASTM E2500 and the ISPE Baseline-Guide Commissioning and Qualification terminology gamp 5 now gives the suppliers a key role in the development processes and in the operation processes gamp 5 has simplified the gamp categories, Chapters of the gamp 4 which describes special types of computer systems were canceled BUT.ģ The gamp 5 remains compatible with the principles in gamp 4 Slide 61.
GAMP 4 AND GAMP 5 GUIDELINES. DRIVERS
gamp 4 and gamp 5 differences Why revision of gamp 4: To align with the concepts and terminology of recent regulatory and industry developments such as:-ICH Q 9 (Annex 20)-FDA: Pharmaceutical cGMPs for the 21st Century: A Risk based Approach Avoid duplication of activities Leverage supplier activities to the maximum possible extent, while still ensuring fitness for intended use Scale all life cycle activities and associated documentation according to risk, complexity and novelty Recognize that most computerized systems are now based on configurable packages, many of them networked Slide 41.Ģ gamp 4 and gamp 5 differences Drivers for gamp 5 Slide 51. 2 Table of 4 and gamp 5 Prospective Qualification retrospective Qualification strategy Slide 31. CHRIST retrospective Qualification strategyġ gamp 4/5 Prospective / retrospective Qualification of water treatment plants)P. CHRIST prospective Qualification strategy 3. gamp 4 and gamp 5 differences Why revision of gamp 4: To align with the concepts and terminology of recent regulatory and industry developments such as:-ICH Q 9 (Annex 20)-FDA: Pharmaceutical cGMPs for the 21st Century: A Risk based Approach Avoid duplication of activities Leverage supplier activities to the maximum possible extent, while still ensuring fitness for intended use Scale all life cycle activities and associated documentation according to risk, complexity and novelty Recognize that most computerized systems are now based on configurable packages, many of them networked Slide 41. Gamp 4/5 Prospective / retrospective Qualification of water treatment plants)P. Example: bachelor of science Search GAMP 4/5 Prospective / retrospective Qualification …

